Do you have to be intelligent to be creative? Can you learn to be more creative? In this episode, we speak with neuropsychologist Rex E. Jung, PhD, who studies intelligence, creativity and brain function. He discusses why – even if it sounds counterintuitive – intelligence and creativity may not have all that much in common.
Transcript of an interview with Audrey and Rex Jung from the APA website.
Audrey Hamilton: Do you have to be intelligent to be creative? Can you really learn to be more creative? In this episode, we speak with one neuropsychologist who studies intelligence, creativity and brain function. He talks about why – even if it sounds counterintuitive – intelligence and creativity may not have all that much in common. I’m Audrey Hamilton and this is “Speaking of Psychology.”
Rex Jung is an assistant professor of neurosurgery at the University of New Mexico and a practicing clinical neuropsychologist in Albuquerque. He studies both brain disease and what the brain does well – a field of research known as positive neuroscience. His research is designed to relate behavioral measures, including intelligence, personality and creativity to brain function and structure. He has published research articles across a wide-range of topics including traumatic brain injury, lupus, schizophrenia, intelligence and creativity. Welcome, Dr. Jung.

Rex Jung: Thank you, Audrey.
Audrey Hamilton: Could you first of all explain neuroimaging and tell our listeners how it helps researchers understand how people think and act?
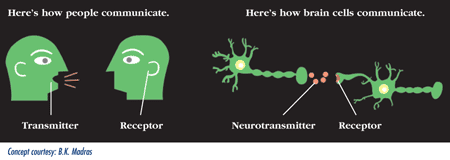
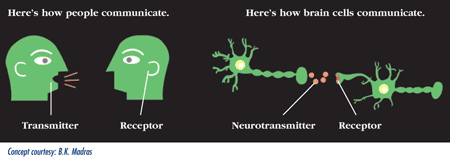
Rex Jung: Sure. So, neuroimaging is the tool that we use to measure the brain and there’s lots of different neuroimaging techniques. I use three main neuroimaging techniques – the first that I learned in graduate school was magnetic resonance microscopy, which sounds kind of complicated. But, it is a technique that basically looks at the chemicals in your brain. It’s in a standard MRI machine like you would go to get your knee scanned. But, using some sophisticated techniques you can look at certain chemicals in the brain. Some of those chemicals are very involved in important neuronal processes. And we’ve correlated those with behavior.
A different technique is called diffusion tensor imaging, which allows us to look at water movement in the brain. And this is important because there’s lots of tubes going through your brain like the wires that connect up your computer to the Internet. And these tubes, called axons, are connecting up different processing modules of your brain and those have to be healthy. So, we can look at the health of those axons, those myelinated axons, the fatty sheath like the insulation that surrounds those tubes.
The third technique that we use is just structural magnetic resonance imaging and that allows us to look at the processing modules of the brain – the cortical thickness – the computers that are on the surface of the brain and how much or little of that you have on the surface of the brain. Those are the three main techniques that I use. There’s functional imaging, fMRI, that most people have heard of where you’re looking a blood flow, as well. Those are ways that we measure brain structure and function and this gives us the ability to do scientific measures that then we can correlate to behavioral measures in psychology.
Audrey Hamilton: Does being highly creative mean you’re also more intelligent?
Rex Jung: Not necessarily. There’s a controversy about this in the psychological literature and some people have found correlations between creativity and intelligence. They’re usually pretty low, this association. And some people make a lot of that, this low association. But usually, because this association between creativity and intelligence is low, it means that you don’t necessarily have to be intelligent to be creative. So, I spent over a decade studying intelligence. It’s one of the reasons I started studying creativity because it seemed like something distinctly different and interesting than intelligence, which I have studied. I work with very highly intelligent people in academia and scientists and not all of them are creative. Why is that? If they do go together I would be working with all of the creative people in my city in Albuquerque, but that wasn’t the case so creativity seemed to be something different.
Audrey Hamilton: Can a person learn to become more creative or simply gain intelligence?
Rex Jung: There are some tools and techniques that can help people to be more creative. We’re starting to learn more about creativity and it’s one of the things that I’m excited about in terms of creativity is that there might be ways to increase your creative capacity.
Intelligence unfortunately seems to be much more under tight genetic control. The genetic correlates of intelligence are high, like .75. So, if you have twins – they’re going to be identical twins – their correlation of their intelligence with one another is going to be very, very high. So that implies that the genetic involvement of that capacity is under much more tight control than the environment would be.
With creativity, we don’t have that information and I’m hopeful that you can modulate or modify creative cognition much more than intelligence. There are studies out there that have shown increases in intelligence scores of two, maybe three points on a particular measure, which are not particularly high. But those are also controversial. Some have been replicated. Some haven’t been replicated. And we really don’t see that in terms of intelligence. With creativity, there’s a pitched effort to try to increase creativity scores on some of these measures and we’re seeing some good initial results and I’m very hopeful about that.
Audrey Hamilton: How does the way a person’s brain works and is structured influence how creative or intelligent he or she is?
Rex Jung: The research that we’ve done shows that the brain organization of intelligence and creativity are quite different. So, when you think about those measures that I talked about, those neuroimaging measures, the brain of someone who is intelligent – think of bigger, better, stronger, faster – all the measures are pointing to higher integrity of the brain of someone who has high intelligence. So, the cortical mantle is thicker, the white matter, the wires are more myelinated, the water can travel faster and in a coherent direction, you have more of these certain chemicals that I was talking about.

Audrey Hamilton: It’s beefed up.
Rex Jung: It’s beefed up, yes. So you can have a better organized brain.
With creativity, the story was different. In different regions of the brain, we were seeing weaker connections, thinner cortex and different levels of these same biochemicals. So, it was really clear from these studies that intelligence and creativity were different because we were seeing different pictures in the measures we were taking of the brain. But I tend to look at creativity and intelligence as two different kinds of reasoning. That creativity is kind of reasoning without all of the information present. So, call it abductive reasoning. But, you have hypothesis testing about how the world could work without all of the information present. So, you have to use abstraction and metaphor and stuff like that about this might look like this or this might be this way.
With intelligence, you’re using deductive reasoning, where it’s rule-based reasoning where a equals b and that’s the way it goes. You have a rule for how this relationship works. So, creativity and intelligence are probably different types of reasoning. Both are very adaptive, but they’re just different for different types of problems that you have to solve out in the world.
Audrey Hamilton: Is real creativity rare? How about genius?
Rex Jung: So, creativity is common and genius is a lot more rare than we would believe. The term genius gets thrown around a lot. But, I think genius is rare because that combination of brain organization where you have high fidelity, beefed up brain in certain regions and then kind of down regulated brain in other regions is really going to be kind of rare where that is present in the same brain. So, to have that back and forth between intelligence and creativity, the ability to do both of those reasoning processes well, where you can do first approximations, hypothesis testing, abstraction and then create a rule, a novel and useful rule out of nothing before is rare and that is true genius.
Audrey Hamilton: Well great. Thank you so much for joining us, Dr. Jung. It’s been very, very interesting.
Rex Jung: Great. Thank you, Audrey.